As its name implies, this particle carries no electrical charge. The other nuclear particle is the neutron. This means that in any atom, the number of protons in the nucleus (often referred to as the nuclear charge) is balanced by the same number of electrons outside the nucleus. Protons are the carriers of positive electric charge in the nucleus the proton charge is exactly the same as the electron charge, but of opposite sign. 
The nucleus is itself composed of two kinds of particles. The radius of an atom must be defined arbitrarily, such as the boundary in which the electron can be found with 95% probability. The latter has no definite outer boundary, so neither does the atom. It is common (but somewhat misleading) to describe the volume of space in which the electrons of an atom have a significant probability of being found as the electron cloud. The extremely small mass of the electron (1/1840 th the mass of the hydrogen nucleus) causes it to behave as a quantum particle, which means that its location at any moment cannot be specified the best we can do is describe its behavior in terms of the probability of its manifesting itself at any point in space. Almost all of the volume of an atom consists of empty space in which electrons, the fundamental carriers of negative electric charge, reside.Almost all of the mass of an atom is contained within a tiny (and therefore extremely dense) nucleus which carries a positive electric charge whose value identifies each element and is known as the atomic number of the element.The most notable of these achievements was Ernest Rutherford's famous 1911 alpha-ray scattering experiment, which established that: The precise physical nature of atoms finally emerged from a series of elegant experiments carried out between 18. Strontium – Properties Summary Element Strontium Atomic Number 38 Symbol Sr Element Category Alkaline Earth Metal Phase at STP Solid Atomic Mass 87.62 Density at STP 2.63 Electron Configuration 5s2 Possible Oxidation States +2 Electron Affinity 5.03 Electronegativity 0.95 1st Ionization Energy 5.6948 Year of Discovery 1790 Discoverer Crawford, Adair Thermal properties Melting Point 777 Boiling Point 1382 Thermal Conductivity 35.3 Specific Heat 0.3 Heat of Fusion 8.\) The Standard English unit is pounds mass per cubic foot ( lbm/ft 3). The standard SI unit is kilograms per cubic meter ( kg/m 3). 
In other words, the density (ρ) of a substance is the total mass (m) of that substance divided by the total volume (V) occupied by that substance. It is an intensive property, which is mathematically defined as mass divided by volume: Typical densities of various substances at atmospheric pressure.ĭensity is defined as the mass per unit volume. How does the atomic mass determine the density of materials? Density of Strontium The atomic mass number determines especially the atomic mass of atoms. The mass number is different for each different isotope of a chemical element. For 63Cu, the atomic mass is less than 63, so this must be the dominant factor. A nucleus with greater binding energy has lower total energy, and therefore a lower mass according to Einstein’s mass-energy equivalence relation E = mc 2. The nuclear binding energy varies between nuclei.This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons. The neutron is slightly heavier than the proton.There are two reasons for the difference between mass number and isotopic mass, known as the mass defect: For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63, and an isotopic mass in its nuclear ground state is 62.91367 u. For other isotopes, the isotopic mass usually differs and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u, since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. 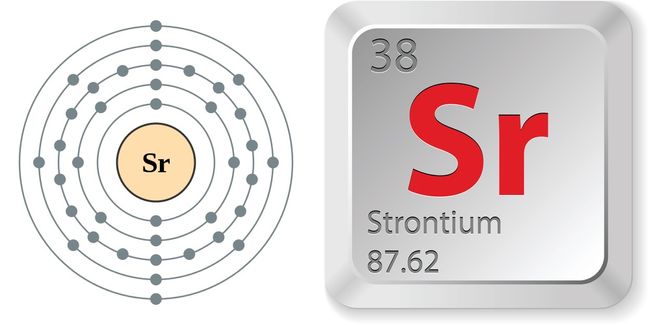
The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Note that each element may contain more isotopes. How does the atomic number determine the chemical behavior of atoms? Atomic Mass of Strontium Since the number of electrons is responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Strontium is a chemical element with atomic number 38 which means there are 38 protons and 38 electrons in the atomic structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
